Your hair feels rough. It tangles constantly. Conditioner slides off without penetrating. You’ve tried every product on the shelf, but nothing works for more than a day. Here’s what’s actually happening: mineral ions from hard water have bonded to your hair shaft at the molecular level, and regular shampoos can’t remove them because they’re not designed to.
This article contains affiliate links. See our affiliate disclosure for details.
Chelation is the chemical process that reverses this damage. It’s not a marketing term or a trendy ingredient claim. It’s a specific molecular mechanism where chelating agents bind to metal ions and make them water-soluble enough to rinse away. Understanding this chemistry explains why some products work and others don’t, and why hard water damage in the Gulf region requires a different approach than what works in soft water areas.
We’re going to break down the science of chelation at the molecular level, explain how different chelating agents work, and show you what actually happens when minerals bond to your hair. This isn’t surface-level cleaning. It’s structural restoration.
What Chelation Actually Means (The Chemistry)
Chelation comes from the Greek word ‘chele’ meaning claw. That’s exactly what chelating molecules do. They grab onto metal ions using multiple binding sites simultaneously, forming a stable ring structure called a chelate complex. This is fundamentally different from how regular surfactants work.
Regular shampoo surfactants (sodium lauryl sulfate, cocamidopropyl betaine) are designed to remove oils and dirt through emulsification. They surround oil molecules and suspend them in water so they rinse away. But mineral ions like calcium (Ca²⁺) and magnesium (Mg²⁺) aren’t oils. They’re positively charged particles that form ionic bonds with the negatively charged sites on your hair’s keratin protein structure.
You can’t emulsify an ionic bond. You need a molecule that can out-compete your hair for those mineral ions. That’s what chelating agents do. They offer more binding sites and stronger attraction than your hair’s protein structure, so the minerals release from your hair and bind to the chelating agent instead. Once bound, the entire complex becomes water-soluble and rinses away.
The most common chelating agents in hair products are EDTA (ethylenediaminetetraacetic acid), citric acid, and sodium gluconate. A 2004 study in the Journal of Cosmetic Science demonstrated that EDTA can remove up to 95% of calcium deposits from hair in a single treatment, compared to less than 15% removal with standard surfactants alone.
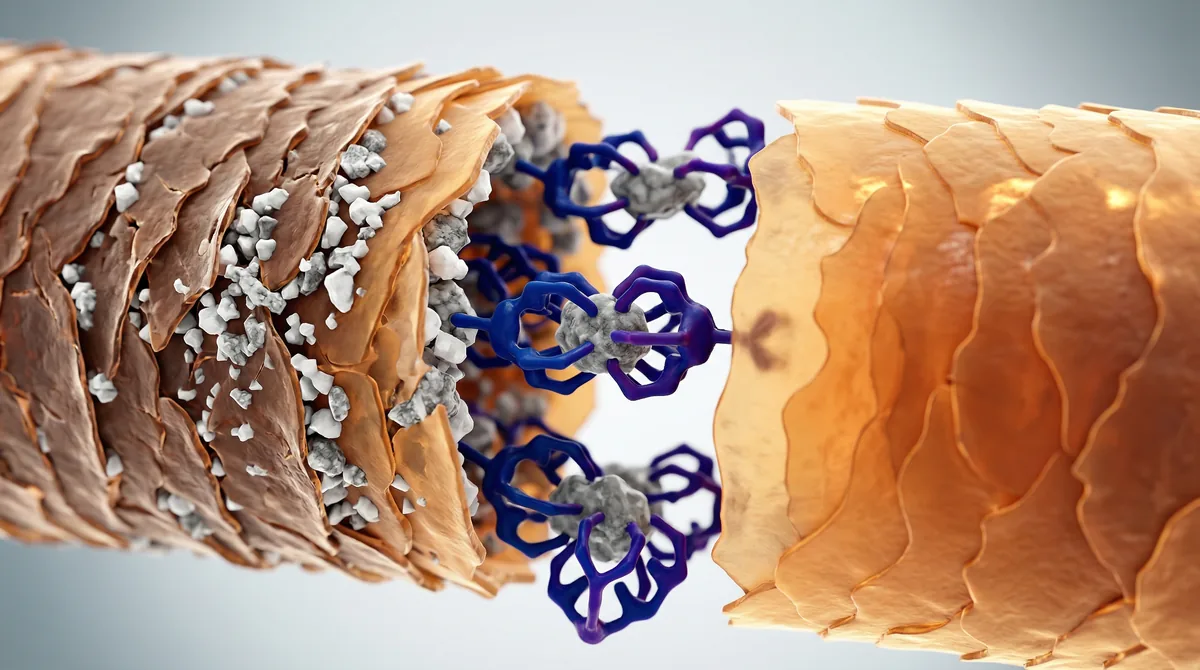
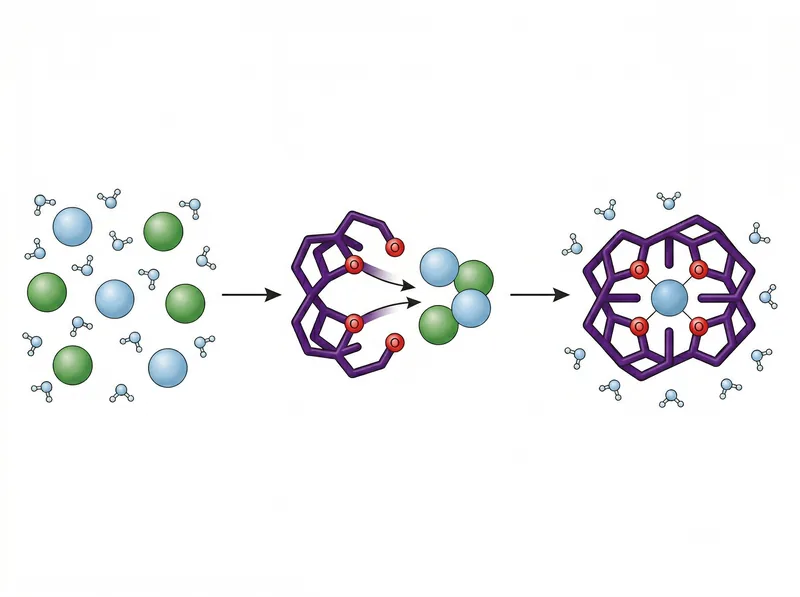 Chelating agents like EDTA use multiple binding sites to form stable, water-soluble complexes with mineral ions, allowing them to be rinsed away.
Chelating agents like EDTA use multiple binding sites to form stable, water-soluble complexes with mineral ions, allowing them to be rinsed away.
How Minerals Bond to Your Hair (And Why They Won’t Let Go)
Your hair shaft has a negative surface charge due to the carboxyl groups in keratin. In soft water, this isn’t a problem. But hard water in the Gulf region contains 200-500 ppm of dissolved minerals, primarily calcium carbonate and magnesium sulfate. When hard water contacts your hair, those positively charged mineral ions are immediately attracted to the negatively charged protein sites.
The binding happens in layers. First, a monolayer of calcium and magnesium ions attaches directly to the hair surface. Then, because these minerals are still partially hydrated, they attract more mineral ions from the water, building up in successive layers. This is why hard water damage is cumulative. Every shower adds another layer.
These mineral deposits do three things that destroy your hair’s appearance and function. One, they physically wedge between cuticle scales, forcing them to lift and separate. Two, they create a rough, irregular surface that scatters light instead of reflecting it, making hair look dull. Three, they increase the hair’s negative charge even further, causing strands to repel each other and creating that characteristic flyaway, frizzy texture.
The worst part? The minerals are hygroscopic. They pull moisture from the air in humid conditions, making hair feel sticky and heavy. In dry conditions, they create a moisture barrier that prevents hydration from reaching the cortex. You’re stuck with hair that’s simultaneously dehydrated and coated in buildup.
Regular shampoos can’t remove this because surfactants don’t break ionic bonds. You need a molecule specifically designed to chelate metal ions. That’s not optional chemistry. It’s the only mechanism that works.
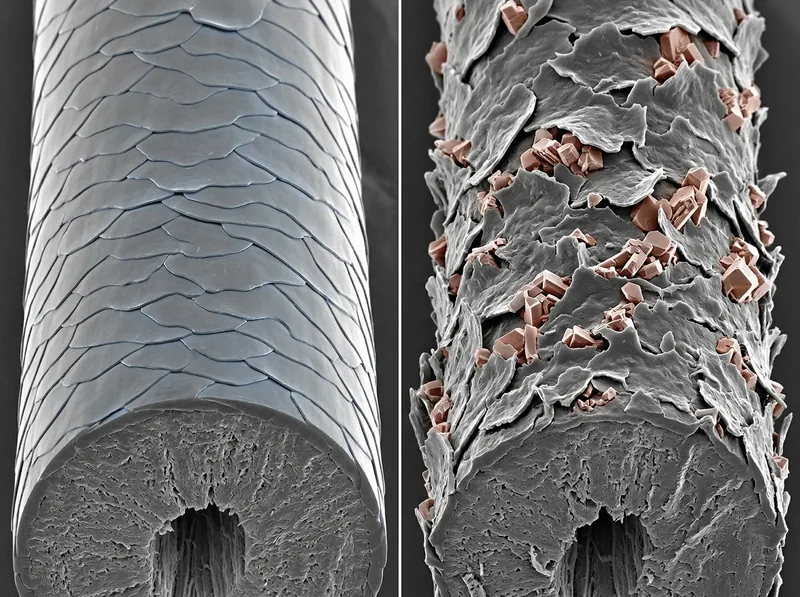 Mineral deposits force cuticle scales to lift and separate, creating a rough texture that catches light poorly and tangles easily.
Mineral deposits force cuticle scales to lift and separate, creating a rough texture that catches light poorly and tangles easily.
EDTA: The Gold Standard Chelating Agent
EDTA (ethylenediaminetetraacetic acid) is the most effective chelating agent used in hair care products. It’s a synthetic amino acid with four carboxyl groups and two amine groups, giving it six potential binding sites for metal ions. This hexadentate structure (six-toothed, in chemistry terms) means EDTA can completely surround a metal ion and form an extremely stable complex.
The stability constant for the EDTA-calcium complex is 10.7 (log K value). For context, anything above 8 is considered a strong chelate. The EDTA-magnesium complex has a stability constant of 8.7. These numbers mean that once EDTA binds to calcium or magnesium, the bond is strong enough that the metal won’t release back onto your hair, even when exposed to more hard water during rinsing.
EDTA works across a wide pH range (4-12), but it’s most effective in slightly alkaline conditions (pH 8-10). This is why many chelating shampoos have a higher pH than regular shampoos. The alkalinity helps open the cuticle slightly, allowing the EDTA molecules to penetrate between the scales where minerals are lodged. Research published in Colloids and Surfaces showed that EDTA penetration into the hair shaft increases by 40% when pH is raised from 5.5 to 9.0.
There’s a common concern about EDTA safety. The Cosmetic Ingredient Review (CIR) panel has evaluated EDTA extensively and concluded it’s safe for use in cosmetics at concentrations up to 2%. The Personal Care Products Council notes that EDTA has been used in personal care products for over 50 years with an excellent safety record. It’s also biodegradable under aerobic conditions.
The only real limitation of EDTA is that it requires adequate contact time. The chelation reaction isn’t instantaneous. It takes 3-5 minutes for EDTA to fully bind to mineral deposits and form stable complexes. This is why chelating shampoos need to be left on the hair, not immediately rinsed.
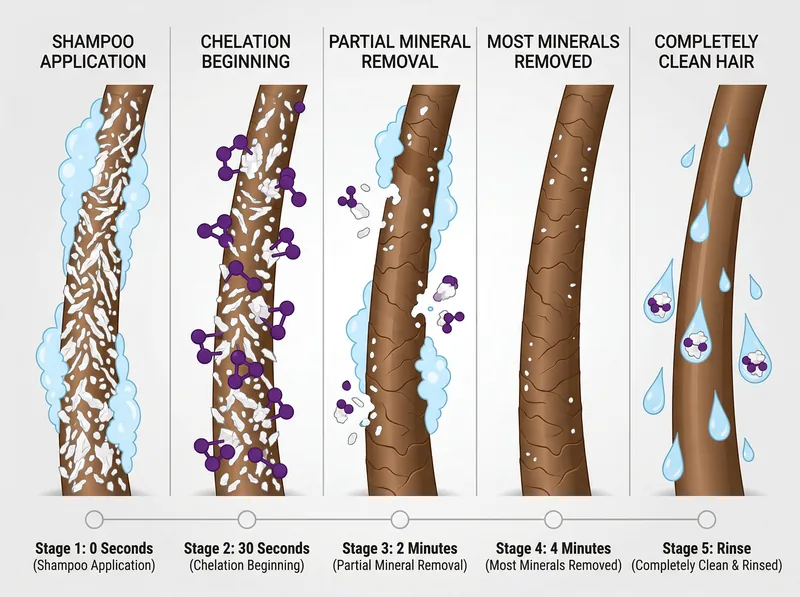 Chelating agents work progressively: binding begins within seconds, but complete mineral removal requires 3-5 minutes of contact time.
Chelating agents work progressively: binding begins within seconds, but complete mineral removal requires 3-5 minutes of contact time.
Alternative Chelating Agents (And When They Work)
Citric acid is the most common natural alternative to EDTA. It’s a weak organic acid with three carboxyl groups, making it a tridentate chelating agent. It can bind to calcium and magnesium, but the stability constants are much lower than EDTA (3.5 for calcium, 3.2 for magnesium). This means the chelate complexes are weaker and more likely to release the minerals back onto your hair.
Citric acid works best as a mild chelator for light mineral buildup or as a pH adjuster that provides some chelating action as a secondary benefit. It’s effective in soft to moderately hard water (below 150 ppm), but in the high mineral concentrations found in Gulf water supplies, citric acid alone isn’t strong enough to remove heavy deposits.
Sodium gluconate is another alternative. It’s derived from glucose and has five hydroxyl groups that can bind to metal ions. The stability constants fall between citric acid and EDTA (4.6 for calcium). It’s gentler than EDTA but also slower-acting. Sodium gluconate is often used in leave-in treatments where extended contact time isn’t an issue.
Phytic acid (inositol hexaphosphate) is a plant-derived chelator found in grains and legumes. It has six phosphate groups that can bind to multiple metal ions simultaneously. The stability constants are comparable to EDTA for some metals, but phytic acid is pH-sensitive and loses effectiveness below pH 6. It’s rarely used in shampoos because of this pH limitation.
For Gulf residents dealing with severe hard water (300-500 ppm), EDTA remains the most reliable choice. The weaker chelators simply can’t compete with the high mineral concentrations. A chelating shampoo like Regrowth+ uses EDTA as the primary chelating agent specifically because of its superior binding strength and stability in high-mineral environments.
The Chelation Process Timeline (What Happens and When)
Understanding the timeline helps explain why technique matters as much as product choice. Chelation happens in stages, and each stage requires time.
0-30 seconds (initial binding): When chelating shampoo contacts your hair, the chelating agents immediately begin binding to the most accessible mineral deposits on the outer cuticle surface. This is the easiest fraction to remove because the minerals haven’t fully integrated into the hair structure yet. You’ll notice the shampoo feels different than regular shampoo during this phase. It doesn’t foam as much because the chelating agents are busy binding to minerals instead of creating bubbles.
30 seconds to 2 minutes (penetration): The chelating agents work their way between the cuticle scales, reaching the mineral deposits lodged in the gaps. The slightly alkaline pH helps here by causing mild cuticle swelling, which creates more space for the chelating molecules to penetrate. This is why you need to work the shampoo through your hair thoroughly, not just apply it to the scalp.
2-5 minutes (deep chelation): This is where the heavy lifting happens. The chelating agents are now binding to minerals that have been building up for weeks or months. These deposits are more tightly bound and require more time to dislodge. The chelate complexes are forming and becoming water-soluble. If you rinse too early, you’ll remove some minerals but leave the deeper deposits intact.
Rinse phase (removal): As you rinse, the water-soluble chelate complexes wash away. The water itself isn’t removing the minerals. The chelating agents already did that work. The rinse just carries away the complexes. This is why even hard rinse water won’t immediately re-deposit minerals. The chelating agents have made them too stable to release.
Most people rinse too early. They treat chelating shampoo like regular shampoo, applying it and rinsing within 60 seconds. That removes surface minerals but misses the accumulated buildup. The difference between a 1-minute wash and a 4-minute wash is the difference between 30% mineral removal and 90% mineral removal.
Why Your Hair Feels Different After Chelation
The first time you use a chelating shampoo properly, your hair will feel strange. Not bad, just different. This is because you’re feeling your actual hair texture for the first time in months, without the mineral coating you’ve gotten used to.
Immediately after chelating, hair feels squeaky clean. Some people describe it as ‘stripped.’ What’s actually happening is that the minerals that were adding false texture and weight are gone. Your hair’s true porosity is exposed. If your hair is naturally fine, it will feel lighter and more prone to static. If it’s naturally coarse, it will feel rougher initially because the smoothing effect of the mineral layer is gone.
This is temporary. Once you apply conditioner to chelated hair, you’ll notice it absorbs dramatically better than before. That’s because the cuticle scales can lie flat again without minerals wedging them open, and the conditioner molecules can actually reach the cortex instead of sitting on top of a mineral barrier. Proper conditioning after chelation is essential.
Within 24-48 hours, properly chelated and conditioned hair develops a different kind of smoothness. It’s not the slick, coated feeling of mineral buildup. It’s actual smoothness from cuticles lying flat and reflecting light uniformly. Hair becomes more manageable, holds styles better, and develops natural shine.
The effect lasts longer than regular shampooing too. Because you’ve removed the accumulated mineral deposits, new minerals from subsequent washes have fewer nucleation sites to attach to. You can often go 5-7 days between chelating treatments instead of needing to chelate every wash.
Chelation Frequency (How Often You Actually Need It)
This depends entirely on your water hardness and how often you wash your hair. There’s no universal answer, but there are guidelines based on mineral concentration.
Soft water (0-60 ppm): You probably don’t need chelating shampoo at all. Regular clarifying shampoo is sufficient. Chelating monthly would be overkill and could potentially over-process your hair by removing too many natural minerals.
Moderately hard water (60-120 ppm): Chelate every 2-3 weeks. This prevents significant buildup without over-chelating. You can use regular shampoo between chelating treatments.
Hard water (120-180 ppm): Chelate weekly. At this mineral concentration, buildup becomes noticeable within 7-10 days. Weekly chelation keeps hair in optimal condition without allowing deposits to accumulate.
Very hard water (180-500+ ppm, typical in the Gulf): Chelate every 3-5 days, or use a chelating shampoo as your primary shampoo. At these extreme mineral levels, buildup happens so rapidly that weekly chelation isn’t sufficient. Some people in the Gulf need to chelate with every wash to maintain hair quality.
You can test whether you’re chelating often enough by paying attention to how your hair behaves. If you notice roughness, tangling, or dullness returning within 2-3 days after chelating, you need to increase frequency. If your hair feels dry or brittle after chelating, you might be overdoing it or not conditioning adequately afterward.
Testing your home water hardness gives you a specific number to work with instead of guessing. A simple TDS meter costs less than two bottles of shampoo and tells you exactly what you’re dealing with.
Chelation vs Clarifying (They’re Not the Same Thing)
This confusion causes people to buy the wrong products and wonder why they don’t work. Clarifying and chelating are different processes that remove different types of buildup.
Clarifying shampoos use strong surfactants (often sulfates at higher concentrations) to remove oils, silicones, styling products, and sebum. They work through emulsification. They’re excellent at removing oily buildup but do nothing for mineral deposits. If you have hard water damage, a clarifying shampoo will clean your hair but leave all the minerals intact.
Chelating shampoos contain chelating agents (EDTA, citric acid, sodium gluconate) specifically designed to bind to and remove metal ions. They also contain surfactants for general cleaning, but the chelating agents are what make them different. They remove minerals that clarifying shampoos can’t touch.
Some products claim to be both clarifying and chelating, and they can be if they contain both strong surfactants and effective chelating agents. But many ‘clarifying’ shampoos marketed for hard water only contain weak chelators like citric acid, which isn’t strong enough for Gulf water conditions.
The practical difference: If your hair feels coated, heavy, or greasy, you need clarifying. If your hair feels rough, tangles easily, or looks dull despite being clean, you need chelating. If you have both issues (common in the Gulf due to hard water plus humid conditions), you need a product that does both, or you need to use them in sequence.
References
- Safety Assessment of EDTA and Related Ingredients as Used in Cosmetics - Cosmetic Ingredient Review
- Chelating Agents for Calcium Removal from Hair - Journal of Cosmetic Science
- Effect of pH on EDTA Penetration in Hair - Colloids and Surfaces B: Biointerfaces
- Hard Water Effects on Hair and Skin - International Journal of Trichology